After validating muscle weakness and motor deficits in young 6neo mice as model of Pompe disease, our team now evaluated acid α-glucosidase (GAA) enzyme activity to validate enzyme reduction and glycogen substrate levels in non-neuronal and neuronal tissue of this lysosomal storage disease mouse model. Quadriceps femoris muscle and cortical tissue of Pompe 6neo mice were thus analyzed for GAA and glycogen levels using a 4-MUG based GAA assay and a commercially available glycogen assay kit (Sigma, MAK016-1KT), respectively. At the age of 24 weeks, 6neo mice present highly decreased GAA activity in the quadriceps femoris muscle and the cortex (Fig. 1A and B, respectively), validating the value of this mouse model for Pompe disease drug development.
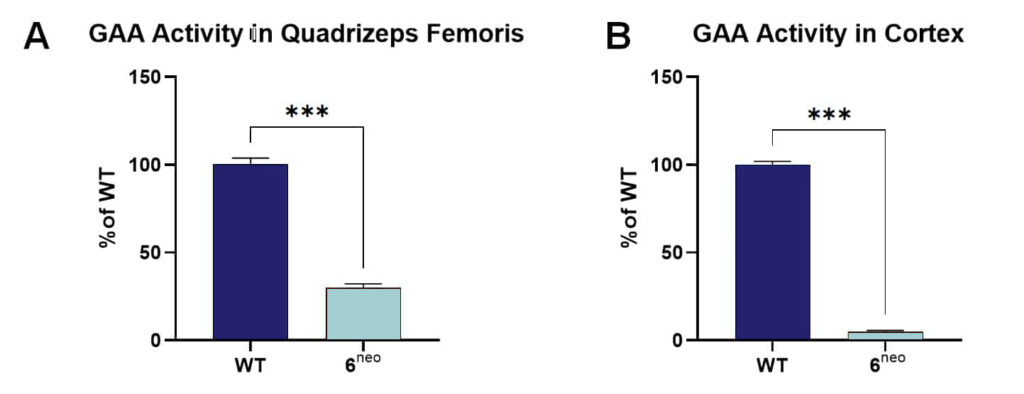
Figure 1: α-glucosidase (GAA) activity in quadriceps femoris muscle and cortex of 6neo mice. Quadriceps femoris muscle (A) and cortical tissue (B) of 24 weeks old 6neo mice were analyzed for GAA activity. n = 16 per group; mean + SEM; unpaired t- test; ***p<0.001.
Already at the age of 4 weeks, glycogen levels are highly elevated in the quadriceps femoris muscle of 6neo mice. With age, glycogen levels further increase (Fig. 2A). Evaluation of glycogen levels in the cortex revealed no significant changes at the age of 4 weeks but highly increased glycogen levels at the age of 24 weeks that further amplify with age (Fig. 2B). Substrate accumulations thus start earlier in non-neuronal tissue.
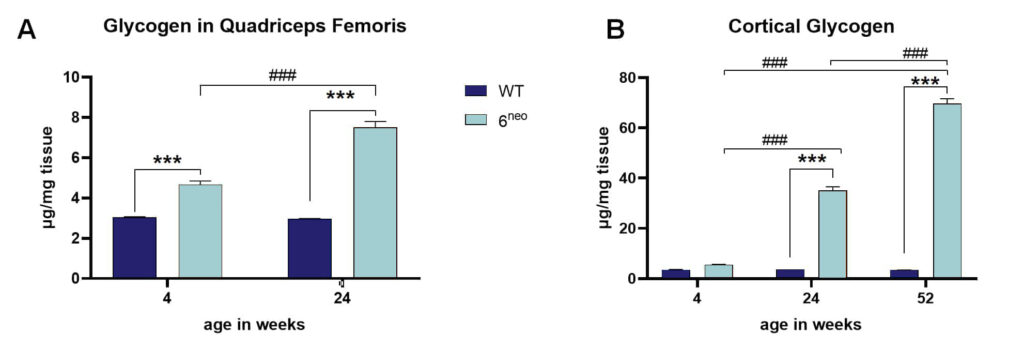
Figure 2: Glycogen levels in quadriceps femoris muscle and cortex of 6neo mice. Quadriceps femoris muscle of 4 and 24 weeks old 6neo mice (A) and cortical tissue of 4, 24 and 52 weeks old 6neo mice (B) were analyzed for glycogen levels. n = 16 per group; mean + SEM; two-way ANOVA followed by Bonferroni’s multiple comparison post hoc test; ***/###p<0.001; *significance between genotypes; #significance between age groups.
As we previously observed first in vivo muscle deficits at the age of 8 weeks, glycogen substrate accumulation seems to proceed in vivo alterations.
These results further promote 6neomice as valuable tool to study Pompe disease and test new compounds for their efficacy to ameliorate the disease’s devastating symptoms.
