Establishment of the CCl4 mouse model allows preclinical testing of your new anti-fibrotic drugs.
Liver fibrosis occurs in most types of chronic liver diseases. In humans, researchers were already able to show that liver fibrosis is reversible, fostering the development of anti-fibrotic drugs. To test the efficacy of these new drugs, proper preclinical models are needed.
Carbon tetrachloride (CCl4) is an organochloride and known as one of the most potent hepatotoxins. As such it is used in preclinical research to induce liver fibrosis in mice. We thus established the CCl4-induced liver fibrosis model to be able to test new drugs against this devastating disease in mice.
C57Bl/6JRccHsd mice at an age of 7 weeks were treated i.p. with CCl4 or vehicle three times per week for a total of 9 weeks. Measurement of body weights at the end of the treatment period showed no significant differences between CCl4– and vehicle-treated animals (Fig. 1A). After sacrifice, liver weights were measured and the liver to body weight ratio was calculated, showing that CCl4-treated animals have significantly increased liver weights compared to controls (Fig. 1B). Quantification of mRNA levels of typical liver fibrosis markers showed highly increased Col1a1 and Acta2 in CCl4-treated mice (Fig. 2A, B). Evaluation of hydroxyproline protein levels by using a hydroxyproline assay as well as Col1a1 and smooth muscle actin (SMA) by immunofluorescent labeling showed a strong increase of these markers in the liver of CCl4-treated animals compared to controls (Fig. 2C-E).
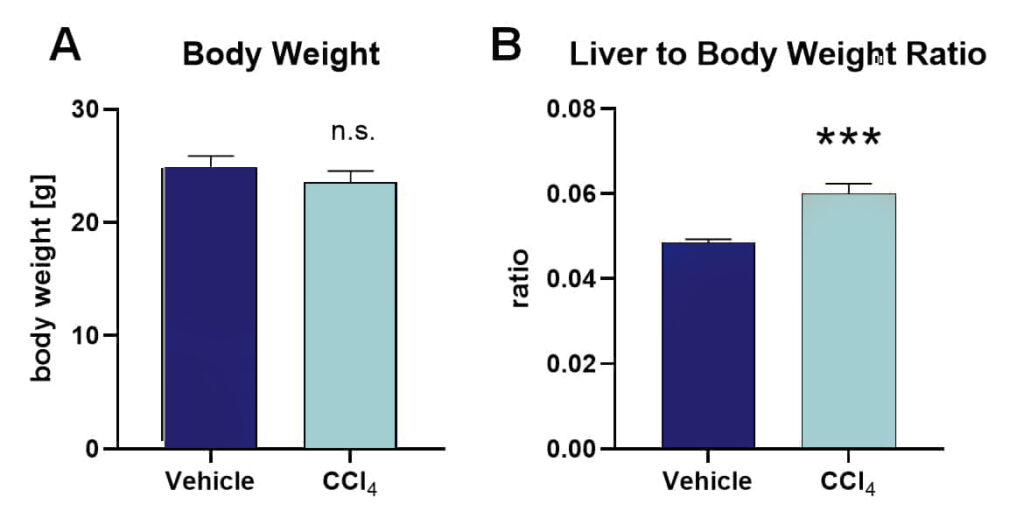
Figure 1: Body weight and liver to body weight ratio of CCl4– and vehicle-treated C57Bl/6JRccHsd mice. A: body weight. B: Liver to body weight ration. Unpaired t-test. n = 6 per group; mean + SEM. ***p<0.001; n.s.: not significant.
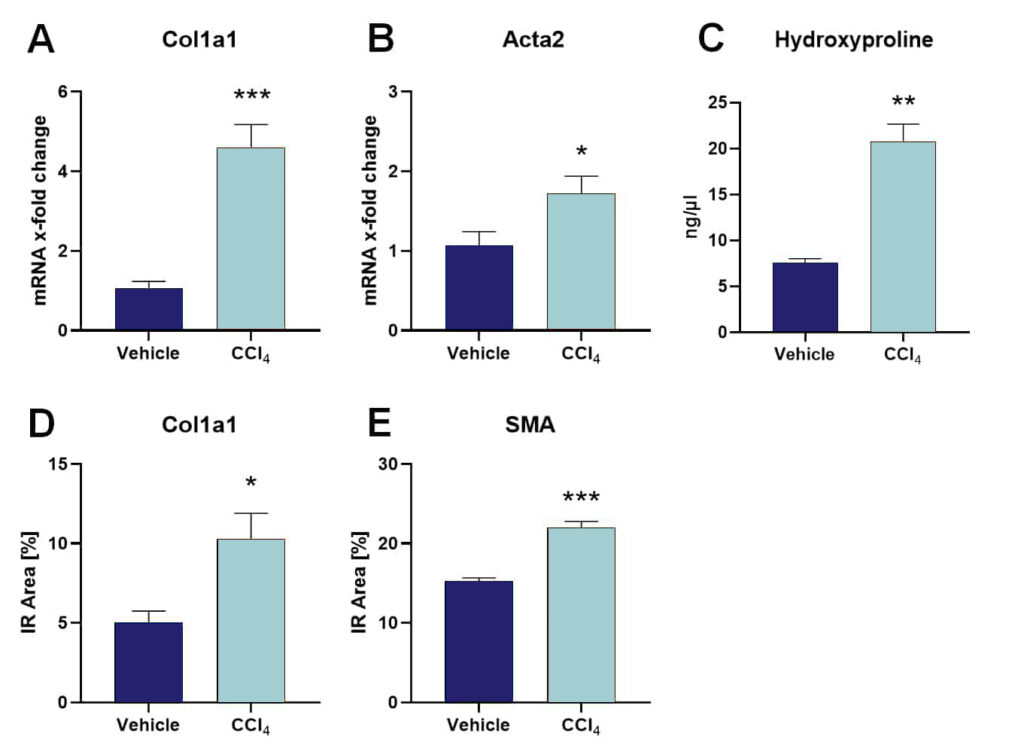
Figure 2: Expression levels of typical liver disease markers after CCl4 treatment. mRNA expression levels of Col1a1 (A) and Acta2 (B). Levels are presented as x-fold change using the 2^(-ΔΔCT) method in relation to the HKG HPRT and Vehicle group. C: Hydroxyproline protein levels in ng/µl. Quantification of Col1a1 (D) and SMA (E) immunoreactive (IR) area in percent. A-E: All markers were measured in the liver after 9 weeks of CCl4 or vehicle treatment. Unpaired t-test; n = 5/6 per group; mean + SEM; *p<0.05, **p<0.01, ***p<0.001.
