Pathological changes in axonal function are well known features of many neurological disorders. Nowadays, stimulation of axonal repair after injury is an important aspect for the development of new therapeutic drugs.
The use of microfluidic chambers (MFCs) can provide unique insights into the axonal compartment independent of the soma. We thus established a model of axonal injury and re- growth in MFCs using dissociated DRG neurons from adult wild type mice. Already 3 days after seeding neurons into the somal side of the MFC, axons start to cross the microgrooves. After 5 days in culture, axons have completely crossed the grooves and built a beautiful network on the axonal side (Fig 1 A). Neurons can then be axotomized by fast removal of the medium from the axonal compartment (Fig. 1 B).
Developmental compounds can be selectively applied to either axonal or somal side alone, by using a hydrostatic pressure difference between the somal and the axonal chamber. The effect of NGF application to the axonal side in comparison to vehicle-treated cells was quantitatively analyzed, showing a significant increase of branching and axonal length, while no impact on the number of crossing axons could be observed (Fig.2).
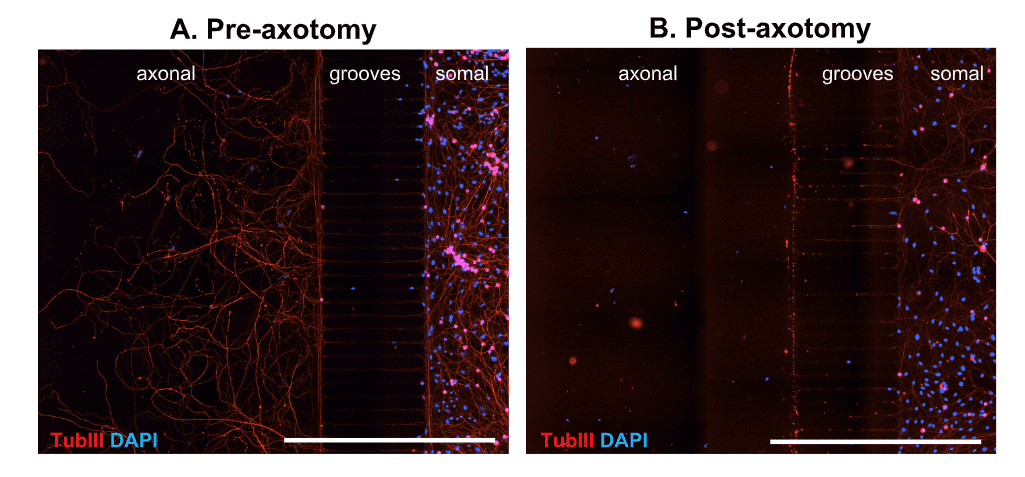
Figure 1. Representative images of DRG neurons from adult wild type mice in microfluidic chambers. DRG neurons were cultured until DIV5 and fixed before (A) and after (B) mechanical axotomy. Tubulin III (TubIII) labeling to visualize neurons and their extensions and DAPI to counterstain for nuclei. Crossing axons on the axonal side before axotomy (A) and removal of axons by axotomy (B). Scale Bar 1,000 µm.
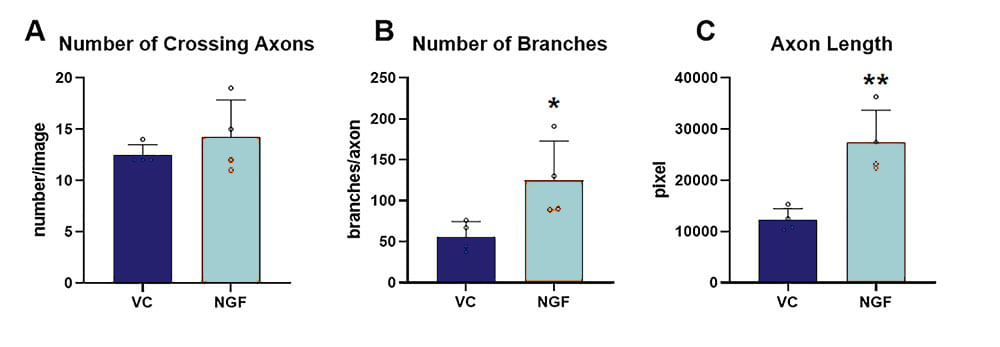
Figure 2. Effect of NGF treatment on axonal re-growth 24 h after axotomy. The following marker were assessed using image-based quantification of total number of crossing axons (A), the number of branch points per crossing axon (B) and the total length of all axons (C); n = 4 wells per group. Unpaired t-test; *p<0.05; **p<0.01. VC: vehicle control; NGF: nerve growth factor.
