zQ175DN-KI mice (JAX# 029928) have the mouse Htt exon 1 replaced by the human Htt exon 1 sequence with ~180-220 CAG repeats. Furthermore, the neo cassette of these knockin mice was deleted (DN). zQ175DN KI mice are thus a model of high CAG repeat Huntington’s disease (HD).
The most important characteristics of zQ175 mice are:
- High mHTT levels in distinct brain regions
- Reduced body weight
- Reduced general health
- Reduced motor skills
mHTT levels are highly increased in the cortex of zQ175 mice at the age of 5.5 months. Similar results can be observed for the caudate putamen. Quantification of mHTT can be performed by sandwich immunosorbent assay (Figure 1). Quantification of human HTT can be performed by immunofluorescent labeling (data not shown).
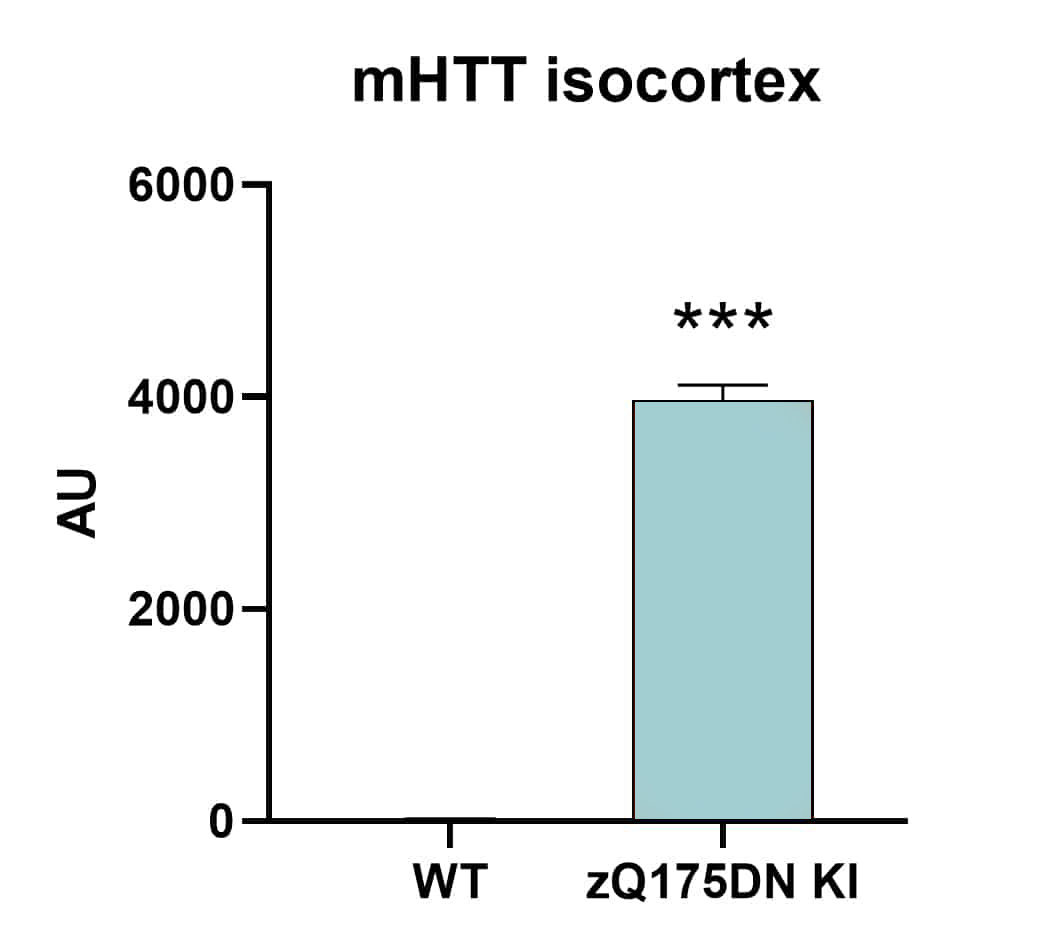
Figure 1: Quantification of mHTT in the isocortex of 5.5 months old mixed sex zQ175DN KI and wild type (WT) mice by sandwich immunosorbent assay using the MesoScale Discovery platform. zQ175DN KI: n = 12; WT: n = 3. Mean + SEM. Unpaired t-test. ***p<0.001.
Evaluation of motor deficits using the beam walk test reveals a strong increase in the number of slips of zQ175 mice at the age of 5.5 months compared to age-matched wild type animals (Figure 2).
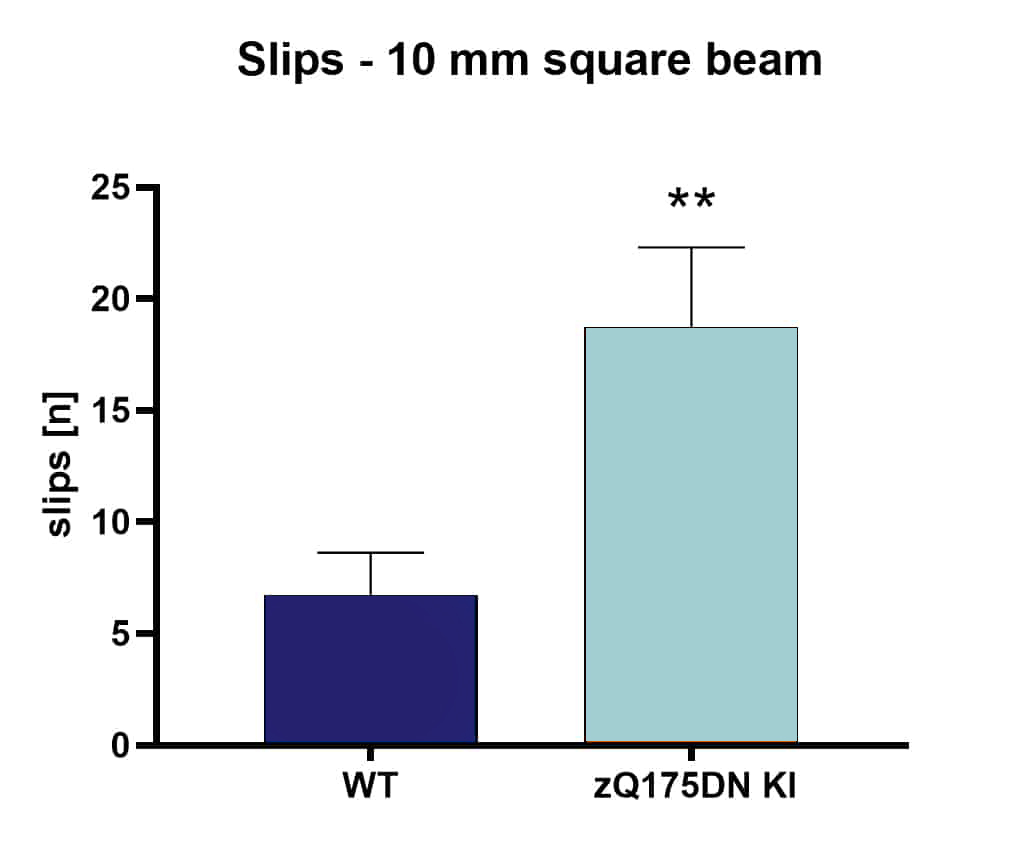
Figure 2: Beam walk test of 5.5. months old mixed sex zQ175DN KI and wild type (WT) mice. Number of slips on a 10 mm square beam. n = 12 per group. Mean + SEM; unpaired t-test. **p<0.01.
Scantox offers a custom-tailored study design for zQ175 mice, and we are flexible to accommodate your special interests. We are also happy to advise you and propose study designs. zQ175 mice show a relevant HD phenotype at the age of 5.5 months. Wild type littermates are available as control animals needed for proper study design.
We are happy to evaluate the efficacy of your compound in the zQ175 mouse model! The most common readouts are:
Looking for something else? Please contact us!
