Excitotoxicity is the pathological process in which neurons are damaged and killed by excessive stimulation by neurotransmitters such as glutamate. This occurs when receptors for the excitatory neurotransmitter glutamate, such as the NMDA receptor and AMPA receptor, are overactivated by a glutamatergic storm. The overstimulation of these receptors leads to an increased calcium concentration in the cells, mitochondrial stress, and a deficiency in autophagy, which finally leads to the death of glutamatergic neurons.
Excitotoxicity is discussed to be involved in stroke, traumatic brain injury as well as neurodegenerative diseases such as multiple sclerosis, Alzheimer’s disease, amyotrophic lateral sclerosis (ALS), Parkinson’s disease, Huntington’s disease, and multiple sclerosis.
In the context of stroke and traumatic brain injury, the abrupt surge of neurotransmitters like glutamate can trigger an excessive influx of calcium ions into the cells, leading among other effects to mitochondrial dysfunction, lysosomal dysfunction, apoptosis and inflammatory processes.
Moreover, excitotoxicity does not only occur in acute injury but also may contribute to neurodegenerative diseases. In multiple sclerosis, immune-mediated inflammation and demyelination are the central pathogenic processes, yet excitotoxicity’s potential role in exacerbating neuronal damage has gained interest. The pathologies of Alzheimer’s disease, ALS, Parkinson’s disease, and Huntington’s disease are as well suspected to be caused by the contribution of excitotoxic processes.
While the extent of involvement of excitotoxicity in each of these conditions varies, therapeutic intervention strategies that are aimed at modulating the balance of neurotransmitters within the central nervous system have potential as possible treatments for patients suffering from these diseases.
At Scantox, excitotoxicity can be assessed by, e.g., treating embryonic primary cortical neurons with glutamate and measuring viability and cytotoxicity (Fig. 1). The substance MK-801 can serve as a reference compound that reverses this lesion.
Glutamate lesions can be performed on SH-SY5Y, NSC-34 or induced pluripotent stem cells (iPSCs). Therefore, cells are treated with glutamate for 24 h. The concentration and time point of glutamate addition depend on the cell type. The glutamate-induced toxicity and cell death are assessed using the LDH and MTT assays.
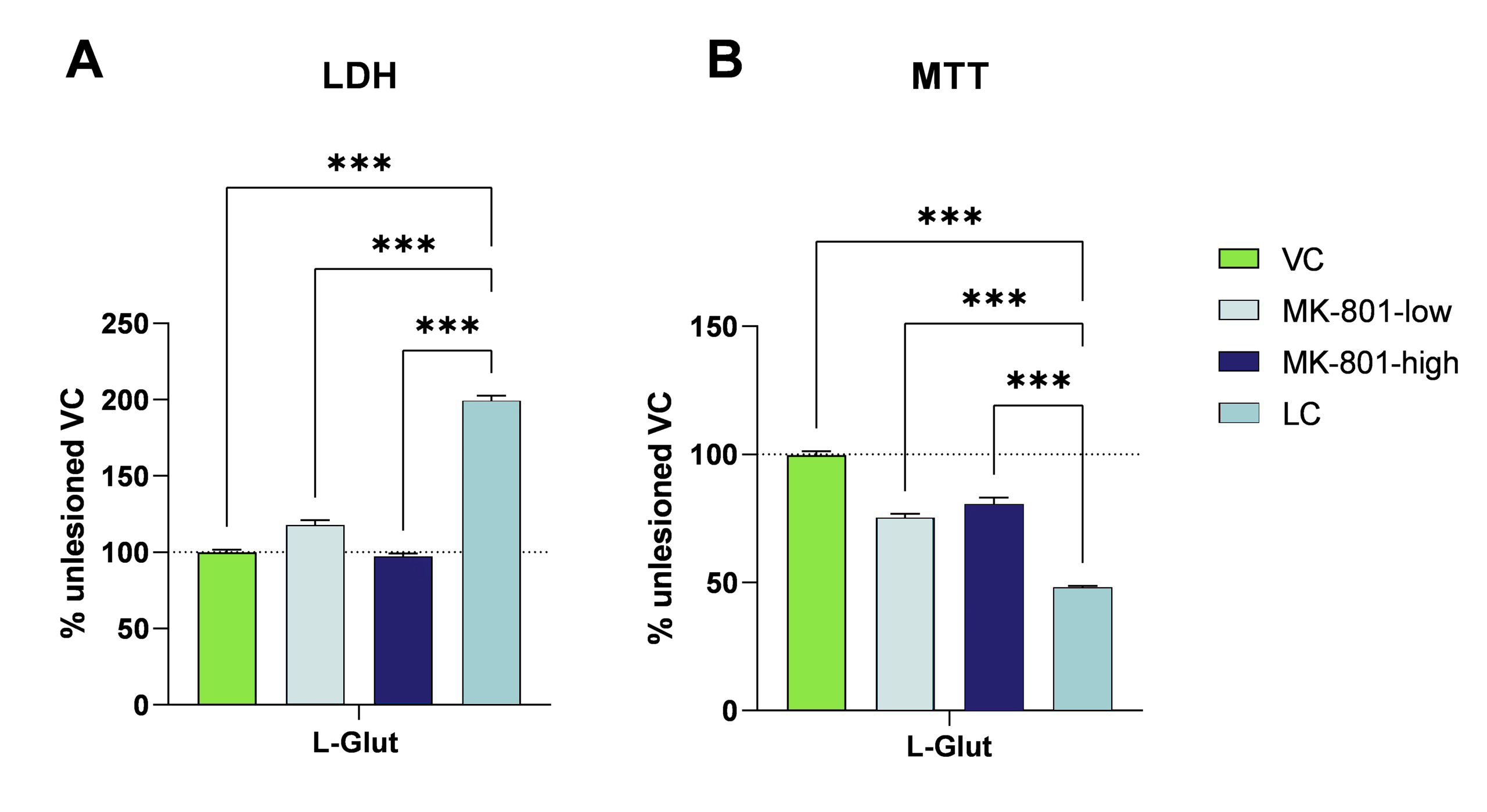
Figure 1: Primary cortical neurons derived from E18 mouse embryos were cultivated until DIV8 and thereafter lesioned with L-glutamate for 24 h. The reference compound MK-801 can restore L-glutamate-induced toxicity and cell death as assessed by LDH and MTT assay, respectively (n=6 per group). Data are displayed as group means and expressed as percent of the vehicle control. One-way ANOVA followed by Dunnet’s post hoc test. ***p<0.001.
