The RNA-binding protein TDP-43 is strongly linked to neurodegenerative diseases like amyotrophic lateral sclerosis (ALS) and frontotemporal lobar degeneration (FTLD). Several studies have shown that cytoplasmic TDP-43 aggregates co-localize with stress granule markers. Stress granules (SGs) are cytoplasmic inclusions that repress translation of a subset of RNAs during cellular stress. Since it was shown that SG formation contributes to accumulation of TDP-43, inhibition of SG formation and/or recruitment of TDP-43 to SGs are pathways that are currently in the focus of ALS research.
To model ALS in vitro, TDP-43-overexpressing neuroblastoma cells can be treated with the well-described SG inducer sodium arsenite (SA). TDP-43 aggregation is then measured by proteinsimple WES technology showing a strong shift from soluble to insoluble TDP-43 species upon SA treatment (Fig. 1).
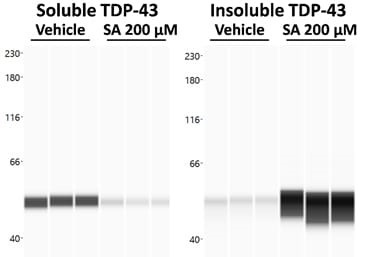
Figure 1. Effect of sodium arsenite (SA) treatment on soluble and insoluble TDP-43 levels. Cells were harvested after SA or vehicle treatment and the soluble and insoluble protein fractions were separated. Both fractions were analyzed for TDP-43 on proteinsimple WES. WES lane view of TDP-43 signal in the soluble and insoluble fraction.
Additionally, SG formation and TDP-43 recruitment can be analyzed by immunocytochemistry (Fig. 2). Immunocytochemical staining for the SG marker G3BP shows substantial SG formation in SA-treated cells compared to the respective vehicle control (Fig. 2), while cycloheximide can counteract SG formation (Fig. 2, bottom row).
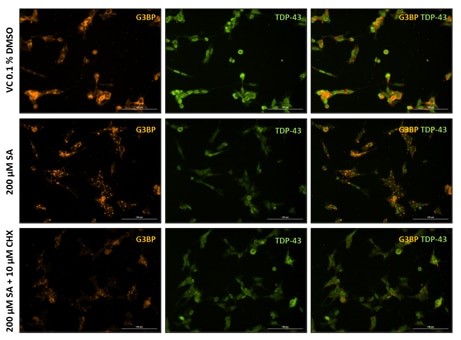
Figure 2. Representative images of vehicle (VC 0.1% DMSO), 200 µM sodium arsenite (SA) and SA lesioned plus 10 µM cycloheximide (CHX) treated SH-TDP-43 cells stained for the stress granule marker G3BP (orange) and human TDP-43 (green). Scale bar 100 µm.
Also changes in TDP-43 localization from nuclear to cytosolic can be assessed using either approach, WES analysis or image-based by immunocytochemistry.
